Platforms
DNA & PlasmidPlatformsRNA Therapeutics PlatformLNP / tLNP Delivery SystemsPolymer & Other Novel Delivery SystemsProtein & Peptide Manufacturing PlatformsGMP Manufacturing & CMC Platforms
Plasmid DNA & IVT Template Manufacturing Platform
A phase-appropriate plasmid DNA CMC and GMP manufacturing platform supporting IVT templates, plasmid raw materials, and DNA drug product programs from early research to clinical and commercial-stage needs.
RNA Therapeutics Viral Vectors & CGT DNA Vaccines / Drugs IVT Template Manufacturing
Built for Plasmid CMC
Vector construction, strain banking, fermentation, purification, analytics, and release testing.
GMP Scale-Up Ready
Flexible plasmid GMP manufacturing from early-stage programs to DNA drug product needs.
Plasmid Platform Proof Points
Platform Scope
Raw Material + DNA DP
Plasmid raw material, IVT template, and DNA drug product manufacturing support.
Process Routes
Supercoiled + Linearized
Manufacturing routes for supercoiled plasmid and linearized IVT template.
Process Design
Flexible Process Routes
Classical three-step, two-step, and one-step process options based on application, timeline, and cost needs.
Template Control
mRNA Template Know-How
Backbone stability, poly(A) integrity, and linearization strategy for IVT template performance.
GMP Scale Proof
1 L–500 L
Flexible plasmid GMP manufacturing scale from development to larger-scale production.
Quality Proof
≥95% scDNA
Supercoiled plasmid ratio target supported by purification and analytical control.
Technical Strengths of CATUG Plasmid Platform
Proprietary Continuous Lysis System
Built in-house and proven for scale-up, CATUG’s continuous lysis platform improves recovery, consistency, and manufacturability for GMP plasmid production.
15+ Years of Plasmid GMP Know-How
Our senior team brings end-to-end experience from plasmid raw materials to commercial DNA drug manufacturing, across scales from 1 L to 500 L.
Regulatory-Ready Method Development
Analytical and quality methods developed with regulatory expectations in mind, supporting impurity control, release testing, comparability, and IND-enabling CMC packages.
Multi-Plasmid Cleaning Validation Excellence
Advanced cleaning validation strategies for multi-plasmid programs, supporting stringent cross-contamination control and GMP manufacturing confidence.
Flexible Process Routes for Cost and Quality
One-step, two-step, and classical three-step purification workflows allow CATUG to balance purity, yield, timeline, and cost based on each product’s use case.
mRNA Template Plasmid Expertise
Beyond plasmid production, CATUG optimizes backbone stability, poly(A) control, linearization, and IVT template quality for RNA drug substance manufacturing.
Supercoiled and Linearized Plasmid Manufacturing Workflow
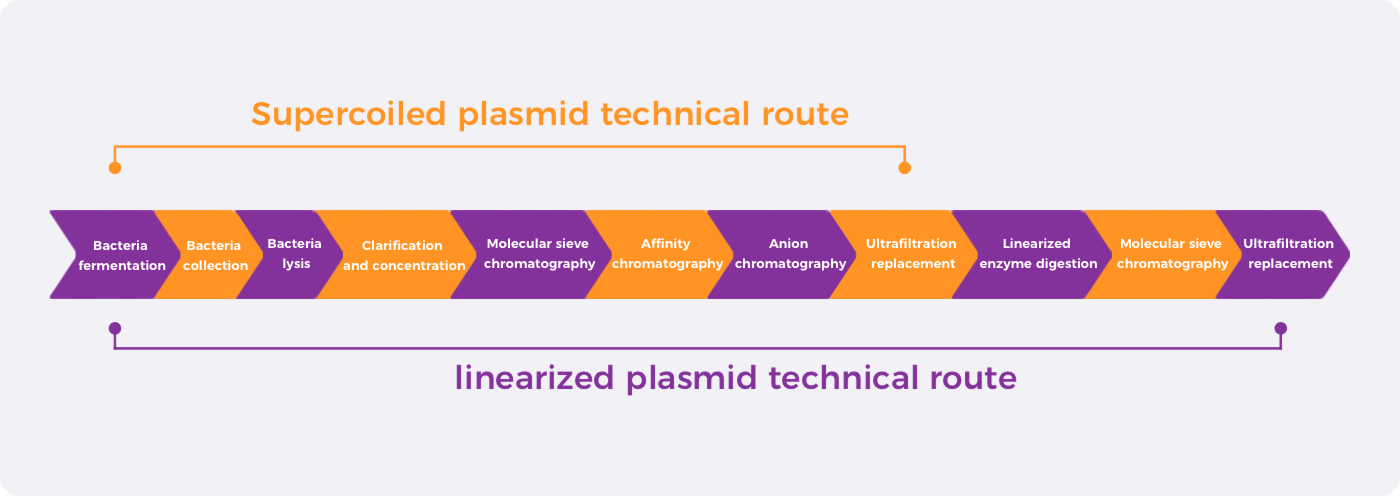
CATUG’s plasmid workflow supports both supercoiled plasmid manufacturing and linearized IVT template preparation, integrating fermentation, lysis, purification, linearization, polishing, UF/DF, and analytical control to support different CMC requirements from early development to GMP manufacturing.
>95%
scDNA Quality Target
Up to 95%
Lysis Step Yield
>95%
TFF Step Yield
99.9%
GMP Linear Template Purity
Supercoiled Plasmid Route
The supercoiled route covers fermentation, harvest, lysis, clarification, chromatography-based purification, UF/DF, and final formulation. It is designed to support high supercoiled plasmid content, impurity clearance, and batch-to-batch consistency for research, pre-GMP, GMP, and DNA drug applications.
Linearized IVT Template Route
The linearized route builds on the supercoiled plasmid process with additional enzymatic linearization, polishing chromatography, UF/DF, and template quality control. It supports linearized template purity, backbone stability, poly(A) integrity, and IVT performance for mRNA, circRNA, saRNA, and gene editing programs.
Stage-Appropriate Plasmid CMC Solutions
From research plasmids to Pre-GMP and GMP/cGMP manufacturing, CATUG delivers phase-appropriate plasmid CMC packages covering process development, analytical control, release testing, stability studies, and IND-enabling documentation.
Research & Construct-Building Package
Vector design, sequence review, cloning, verification, strain screening, and research-grade plasmid preparation for early feasibility studies.
PD Plasmid Development Package
Process development covering fermentation, lysis, clarification, purification, impurity clearance, and scale-up strategy.
IVT Template Optimization for RNA Programs
Plasmid backbone stability, poly(A) integrity, linearization strategy, and IVT template quality control for RNA manufacturing.
Pre-GMP Plasmid CMC Package
Strain bank construction, process protocol, batch record, enhanced QC, stability study, and manufacturing documentation before GMP entry.
GMP Plasmid Raw Material Manufacturing
GMP plasmid raw material manufacturing for RNA therapeutics, viral vectors, gene editing, and gene/cell therapy programs.
Commercial-Level DNA Drug Product
DNA drug product (DP) manufacturing support for DNA vaccines, naked plasmid therapeutics, and DNA-based drug programs from clinical to commercial stages.
Advanced Analytical & Impurity Control
In-house analytical methods for scDNA, purity, topology, endotoxin, HCD, HCR, HCP, residual antibiotics, and stability attributes.
IND-to-Commercial CMC & Tech Transfer
CTD-format CMC packages, batch summaries, analytical reports, regulatory response support, and structured transfer into GMP and commercial manufacturing.
GMP Manufacturing and Tech Transfer Readiness
Explore CATUG GMP Facilities
Take a closer look at CATUG’s plasmid GMP suites, flexible production lines, facility design, quality systems, and tech transfer readiness.
Flexible Manufacturing Configuration
CATUG combines small-scale PCV/IIT lines, single-use systems, and stainless-steel production lines to support fit-for-purpose GMP manufacturing based on project stage, product characteristics, timeline, contamination-control needs, and cost considerations.
GMP Facility Design & Quality Foundation
Our GMP facilities are designed with unidirectional flow and full-air-exhaust principles, supported by quality systems covering QA oversight, batch records, deviation management, change control, release testing, data integrity, and full traceability.
Parallel Execution & Cost-Controlled Scale-Up
Multiple GMP lines enable parallel project execution, flexible scheduling, and efficient scale selection from early development and engineering runs to clinical supply and larger-scale GMP manufacturing.
EU QP-Audited Tech Transfer Readiness
CATUG has successfully passed EU QP audit and maintained a 100% audit pass rate, while supporting structured technology transfer covering process handover, risk assessment, manufacturing readiness, and execution into GMP production.
contact us